

PROTECT‑MI
Clinical Trial
Investigating A New Potential Therapy
for Heart Attack Patients

About the PROTECT‑MI Research Study
PROTECT‑MI is a Phase 2a research study evaluating a potential new therapy aimed at improving outcomes for patients experiencing a severe type of heart attack called ST‑elevation myocardial infarction (STEMI). STEMIs are cause by a blocked artery in the heart, causing heart cells to die.
The standard treatment for STEMI is percutaneous coronary intervention (PCI), This helps by:
Removing the blockage in the heart artery
Placing a small tube (called a stent) to keep the artery open
This restores blood flow to the heart, which is very important.
When blood flow returns suddenly, it can cause extra damage to the heart. This is called reperfusion injury. Right now, there’s no approved treatment to stop this from happening.
What’s the Goal of This Study?
The PROTECT‑MI research study is testing a new potential therapy that may help protect the heart from reperfusion injury following PCI.
The hope is to improve recovery and outcomes for people having a STEMI heart attack.
Who may be suitable?
The research study is enrolling patients who are currently being treated for STEMI.
Eligibility criteria include: Male and female patients aged 40 to 75 years who has not had a prior STEMI.
What is involved in this research study?
If the participant is suitable for the research study:
They will receive the study medication or placebo (inactive substance) through a drip (IV infusion) into a vein for about 6 hours.
They will still receive the standard PCI procedure and all usual care for heart attack.
After leaving the hospital, the participant will be asked to have one follow up by phone and return for one follow-up visits at the study clinic.
All study‑related care, tests, and medication will be provided at no cost.
PARTICIPATION IS VOLUNTARY
Taking part in this research study is completely voluntary. Participants may choose to leave the research study at any time, for any reason.
Why Awareness Matters
Percutaneous coronary intervention (PCI), the standard treatment for STEMI, must be performed within a critical time window-usually within 2 hours of patients arriving in the hospital. In this research study, the study medication will be given at the same time, or shortly after PCI, meaning patients have very limited time to receive information about the trial and provide informed consent.
That’s why early awareness of this research study is essential. Sharing information ahead of time helps ensure that patients, families, and care teams are informed and prepared to consider participation during emergency care.
Where Is It Happening?
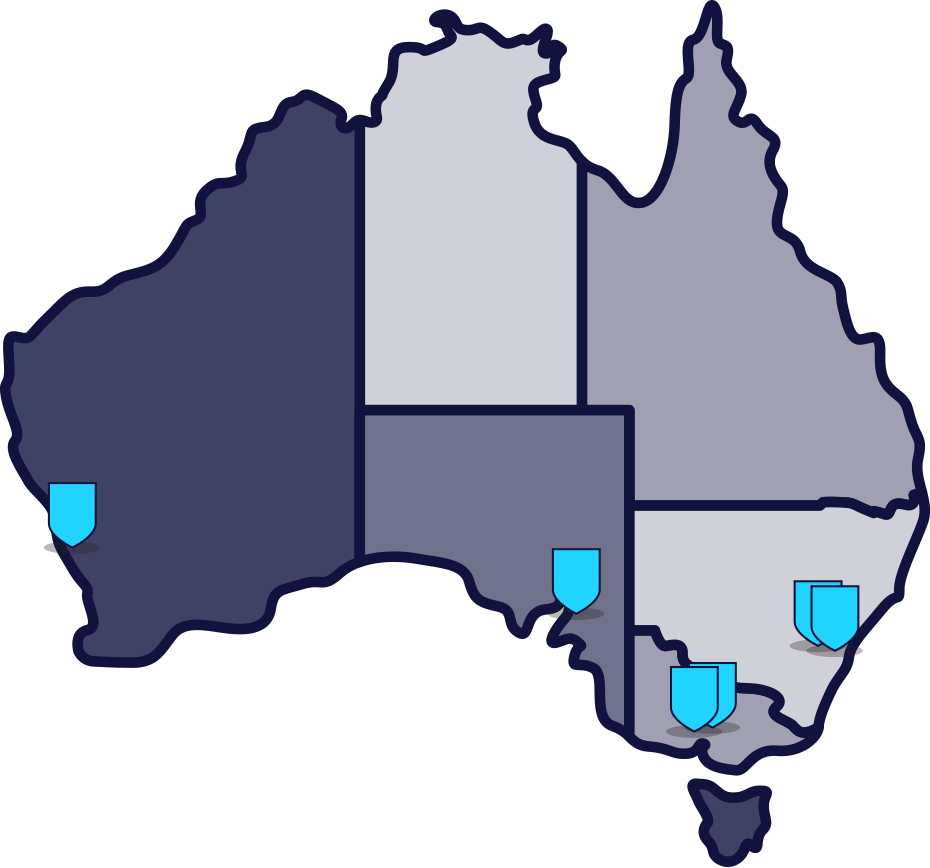
The PROTECT‑MI research study is recruiting at multiple hospitals and research centres across Australia.
Participating Sites:
Sunshine Hospital, VIC
Nepean Hospital, NSW
Royal Adelaide Hospital, SA
Northern Hospital, VIC
Sir Charles Gardiner Hospital, WA
Liverpool Hospital, NSW
Want to Learn More?
For details about the PROTECT‑MI research study protocol and participating sites, please contact:
Name: Sponsor Nyrada
Email: protect-mi@nyrada.com
Phone: +61 2 9498 3390
MEET THE TEAM

Prof. William Chan
Co-ordinating Principal Investigator
PROTECT-MI

Dr. Marissa Lim
Medical Officer
Nyrada Inc

Dr Alexandra Suchowerska
Director Clinical Operations
and Regulatory Affairs
Nyrada Inc
